Who is at risk?
Every treatment decision counts—especially when it comes to reducing the risk of recurrence.
Patients with 1-3 positive nodes and high-risk features have nearly as high a risk of recurrence as those with 4+ positive nodes, yet they are even more frequently undertreated:
A CDK4/6i1DO NOT RECEIVE
A CDK4/6i1
of recurrence.1
These real-world data come from a retrospective study using the US nationwide Flatiron Health EHR-derived de-identified database. Patients with HR+, HER2−, node-positive EBC who underwent breast cancer surgery and then initiated adjuvant ET-based therapy from January 2023 to March 2024 were selected (N=3170). High-risk N1/N1mi disease was defined as Grade 3 or a tumor ≥5 cm. Adjuvant CDK4/6i treatment was assessed from January 2023 through June 2024. Baseline characteristics were summarized descriptively and included nodal status, age, race/ethnicity, insurance, socioeconomic status, and practice setting.1
Results are based on a post hoc subgroup analysis, following the methodology used by Sheffield, et al (2022).2
This analysis was exploratory and did not test a hypothesis.1,2
Reasons for prescribing or not prescribing CDK4/6 inhibitors to eligible patients are not available in the Flatiron Health database. It is unknown which disease characteristics were accessible or considered when making treatment decisions. The Flatiron Health database does not capture comorbidities, financial considerations, or patient and physician discussion.1
WHAT IS THE RISK?
Despite standard ET, patient survival mirrors that of eTNBC3*
Based on real-world evidence, patients with node-positive, high-risk, HR+, HER2– EBC have a mortality rate similar to patients with eTNBC3
- Among both, mortality rate is about 20% by year 53
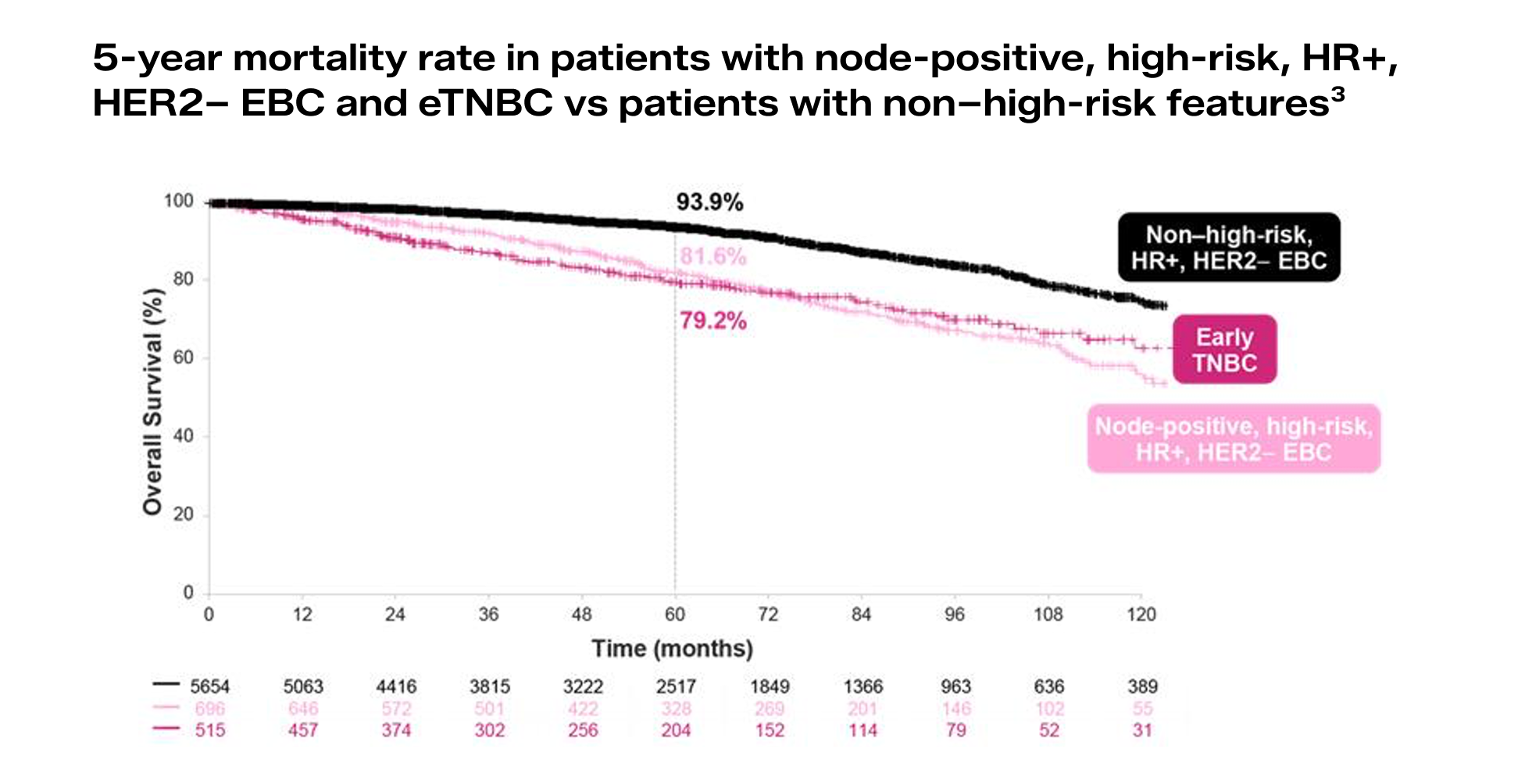
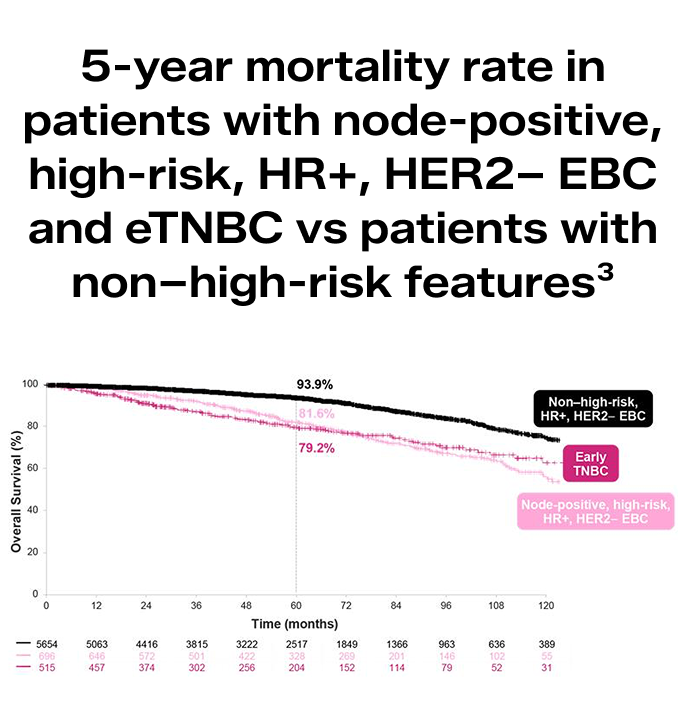
OS was defined as the number of months from start of index treatment to death.3
This was a retrospective real-world evidence study that used the US-based, electronic health record–derived de-identified Flatiron Health Research Database. The analysis dataset included approximately 16,000 patients diagnosed with EBC from January 2011 to June 2024, primarily from the community oncology setting.3
This analysis was exploratory and did not test a hypothesis.3
For the outcomes by risk features, >50% of patients did not have Ki-67 results, and these patients may have been incorrectly classified as non–high-risk, yielding an overestimation of risk for the non–high-risk group and underestimation of relative risk for the node-positive, high-risk groups. Finally, caution should be considered when comparing outcomes for HR+, HER2– EBC and eTNBC, as the analyses for eTNBC began from the start of adjuvant chemotherapy.3
Undertreating patients may mean recurrence isn’t just possible—it’s more likely1,4
Patients with high-risk features are more than 2x as likely to experience recurrence vs patients without those features4
- Risk of recurrence is highest in the first 2 years following primary treatment2
SURVIVAL IS
Making the right choice of adjuvant treatment for these patients is critical:
10-year overall survival for patients with high-risk disease who receive standard adjuvant ET is approximately 60%.1,5
Throughout my whole
ALLEGRA, a real patient diagnosed with HR+, HER2−, high-risk EBC
treatment, I shared I don’t
want to wait for a recurrence.
I want a chance to keep
pursuing my love of travel for
as long as possible.
*Patients with eTNBC had ER–, PR–, HER2– EBC.3


